 A spontaneous change may be so rapid that it is essentially instantaneous or so slow that it cannot be observed over any practical period of time. 
While a catalyst may be used to speed up or slow down a process, its presence does not influence spontaneity: nonspontaneous reactions cannot be made spontaneous using a catalyst. The spontaneity of a process is not correlated to the speed of the process. Spontaneity is Independent of Rate of Reaction At room temperature and typical atmospheric pressure, for example, ice will spontaneously melt, but water will not spontaneously freeze. A process that is spontaneous in one direction under a particular set of conditions is nonspontaneous in the reverse direction. Iron exposed to the earth’s atmosphere will corrode (spontaneous process), but rust is not converted to iron (nonspontaneous process) without intentional chemical treatment. Water will naturally flow downhill (spontaneous process), but uphill flow (nonspontaneous process) requires outside intervention such as the use of a pump. Processes have a natural tendency to occur in one direction under a given set of conditions. A nonspontaneous process, on the other hand, will not take place unless it is “driven” by the continual input of energy from an external source. 
Adding a catalyst, such as manganese oxide, speeds up the reaction and therefore increases the decomposition rate of hydrogen peroxide.Īlthough the catalyst enhances the rate of the spontaneous reaction, it cannot make the nonspontaneous reverse reaction be spontaneous.Ī spontaneous process is one that occurs naturally under certain conditions. The reaction, although spontaneous, proceeds very slowly at room temperature. The reaction could be extremely fast, like an acid-base neutralization, or extremely slow, like the conversion of diamonds to graphite.Ĭonsider the decomposition of hydrogen peroxide into water and oxygen gas. Spontaneity in thermodynamics does not indicate the speed of a process. Similarly, if a hot object is placed in cold water, the heat from the object will be spontaneously transferred to the water until the temperatures of the object and the water reach equilibrium. If a flask containing a gas is connected to another empty flask through a valve, opening the valve will lead to the spontaneous transfer of the gas to the empty flask, which will continue until the gas is equally distributed in both flasks. Spontaneous processes occur due to the dispersal of energy and matter. For example, ice that melts at room temperature cannot spontaneously freeze back to its original form. Spontaneity is affected by conditions such as temperature and pressure.įor example, the melting of ice at a temperature of 0 ☌ or below is not spontaneous however, the melting of ice at room temperature is spontaneous because it does not need any external intervention.Ī spontaneous reaction is usually unidirectional, and its reversal requires an external force to make it proceed as a nonspontaneous reaction. 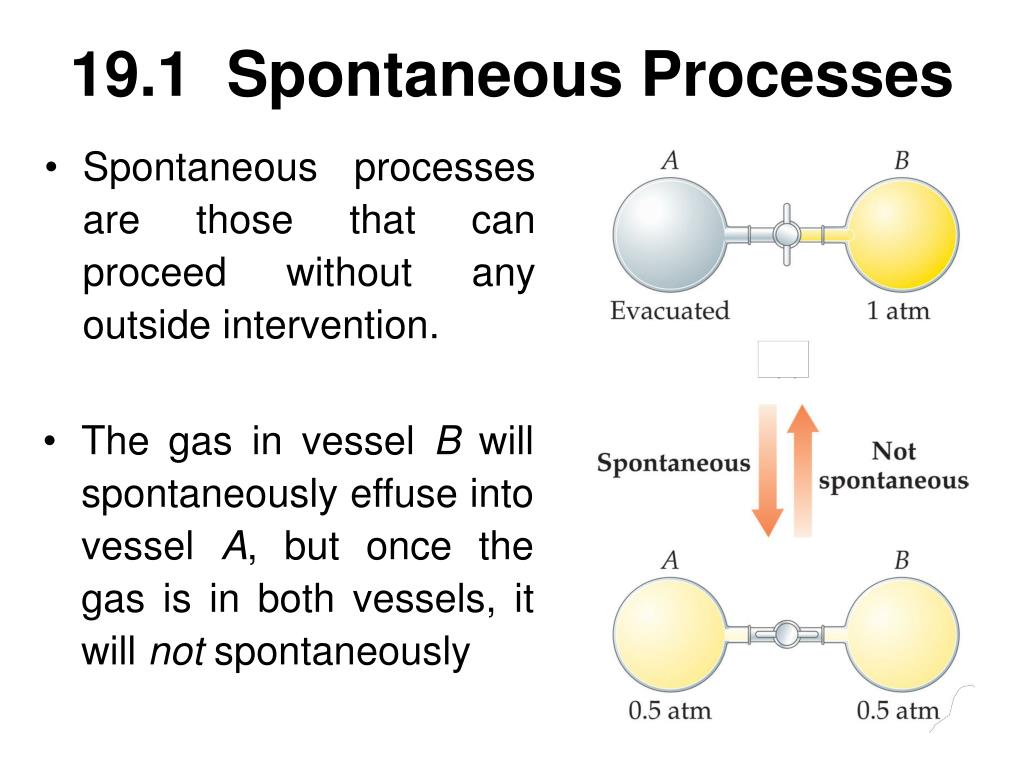
A nonspontaneous process is one that requires external forces to proceed. A spontaneous reaction is one that occurs without any external intervention under a particular set of conditions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
